Global Real-World Evidence (RWE) Market Set to Surpass $10 Billion by 2030
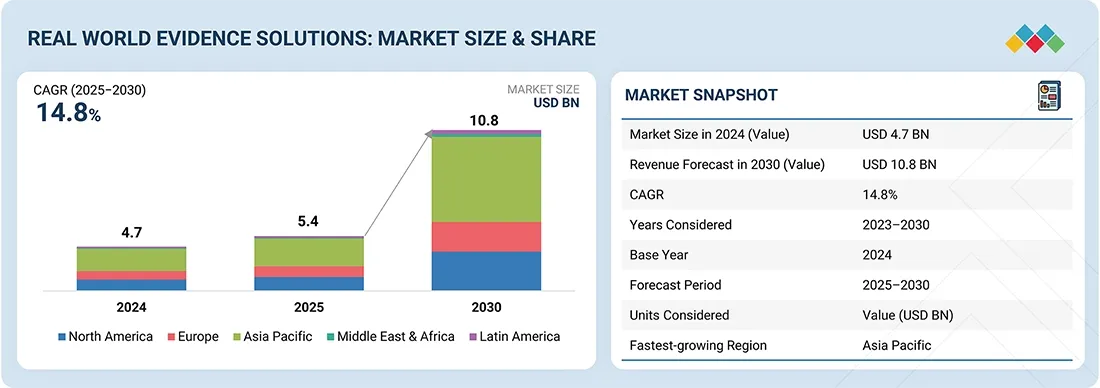
The healthcare landscape is undergoing a fundamental shift as data-driven decision-making moves from controlled clinical environments into the "real world." According to a new market analysis by MarketsandMarkets, the global Real-World Evidence (RWE) solutions market is projected to skyrocket from its 2024 valuation of USD 4.74 billion to a staggering USD 10.83 billion by 2030.
This growth, representing a compound annual growth rate (CAGR) of 14.8%, signals a new era for pharmaceutical R&D, regulatory approvals, and value-based care.
Download PDF Brochure:https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=76173991
A Perfect Storm of Drivers
The surge in RWE adoption is being fueled by a "perfect storm" of demographic shifts and technological advancements. A primary driver is the rising global geriatric population, which has led to an increased prevalence of chronic diseases such as cancer, diabetes, and cardiovascular conditions.
"RWE solutions are becoming essential for healthcare systems to develop personalized, cost-efficient treatment strategies," the report notes. This is further supported by a regulatory environment where agencies like the FDA and EMA are increasingly accepting RWE to support label expansions and post-market safety monitoring.
Market at a Glance: 2025–2030
| Metric | Details |
| Market Size (2030) | USD 10.83 Billion |
| Growth Rate (CAGR) | 14.8% |
| Fastest Growing Region | Asia Pacific (16.0% CAGR) |
| Dominant Revenue Model | Subscription (41.3% share) |
Digital Transformation and AI Integration
The transition to cloud-based solutions is accelerating the market's evolution. In 2024, cloud deployment dominated the sector due to its ability to integrate massive, disparate datasets including:
- Electronic Health Records (EHRs)
- Genomics and Proteomics
- Claims and Pharmacy data
- Wearable device and digital health data
The integration of Artificial Intelligence and Machine Learning (AI/ML) is also identified as a critical trend, allowing researchers to extract meaningful patterns from fragmented data to support precision medicine and faster drug development.
Regional and Competitive Dynamics
While North America currently holds the largest market share—thanks to robust R&D investment and early EHR adoption—the Asia Pacific region is emerging as the fastest-growing market. Countries like China, India, and Japan are benefiting from digital health initiatives and a large patient population that generates substantial real-world data.
The competitive landscape is a mix of established "Star" players and agile innovators. Industry giants such as IQVIA, Optum, and ICON plc continue to lead through scale and global infrastructure. Meanwhile, specialized firms like Aetion and Flatiron Health (recently involved in significant M&A activity with Datavant and Unicancer) are pushing the boundaries of regulatory-grade evidence generation.
Overcoming Challenges
Despite the optimistic outlook, the industry still faces hurdles. A primary restraint is the continued reluctance of some medical practitioners to rely on observational studies over traditional Randomized Clinical Trials (RCTs). Furthermore, the lack of universally accepted methodology standards and a shortage of skilled data professionals remain significant challenges for the global ecosystem.
As the industry moves toward 2030, the ability to ensure data quality and standardization will be the deciding factor in how quickly RWE becomes the primary engine for next-generation healthcare decision-making.