Europe In Vitro Diagnostics Market Size, Share & Growth
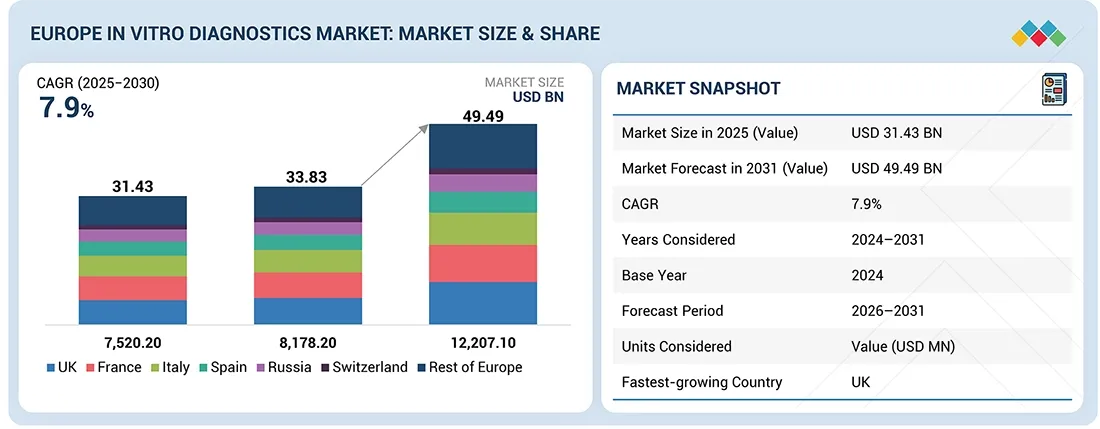
The Europe in vitro diagnostics (IVD) market, valued at USD 31.43 billion in 2025, stood at USD 33.83 billion in 2026 and is projected to advance at a resilient CAGR of 7.9% from 2026 to 2031, culminating in a forecasted valuation of USD 49.49 billion by the end of the period.
Download PDF Brochure:- https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=20552722
- By country, Germany accounted for the largest share of the European in vitro diagnostics market in 2025.
- By product & service, the reagents & kits segment is expected to register the highest CAGR of 8.4%.
- By technology, the immunoassay segment accounted for the largest market share in 2025, while molecular diagnostics is expected to register the highest CAGR during the forecast period.
- By specimen, the blood, serum, and plasma specimens segment is expected to dominate the market.
- By site of testing, the laboratory test segment held the largest share of the market in 2025.
- By application, the infectious diseases segment is expected to dominate the market.
- By end user, clinical laboratories segment will grow the fastest during the forecast period.
- F. Hoffmann-La Roche Ltd., Abbott, Siemens Healthineers AG, Danaher Corporation, and Thermo Fisher Scientific Inc. were identified as some of the star players in the European in vitro diagnostics market, supported by their strong market presence and extensive product portfolios.
The Europe in vitro diagnostics market is experiencing strong growth due to the increasing burden of chronic and infectious diseases, an aging population requiring regular diagnostic monitoring, and expanding preventive healthcare initiatives across the region. Greater awareness of early disease detection, combined with rising participation in routine health screening programs, is improving testing uptake.