Asia Pacific In Vitro Diagnostics Market Size, Share & Growth

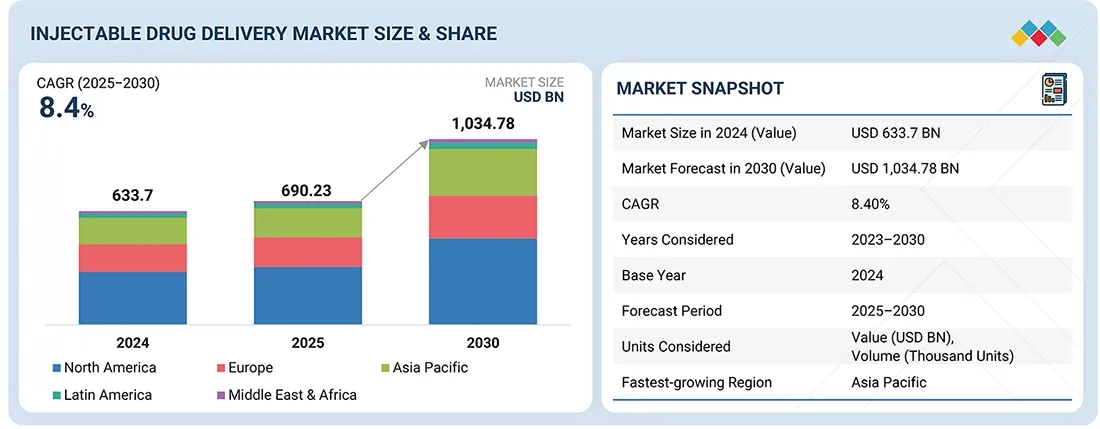
The Asia Pacific in vitro diagnostics market is valued at an estimated USD 29.27 billion in 2026 and is projected to reach USD 45.11 billion by 2031 at a CAGR of 9.0% during the forecast period. Market expansion is being driven by increased access to diagnostic services, resulting from the gradual improvement in the region's healthcare sector infrastructure.
Download PDF Brochure:- https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=90323365
- By country, China accounted for the largest share in the Asia Pacific in vitro diagnostics market in 2025.
- By product & service, the reagent & kits segment is expected to register the highest CAGR of 9.5%.
- By technology, the immunoassay segment accounted for the largest market share in 2025, while molecular diagnostics is expected to register the highest CAGR during the forecast period.
- By specimen, the blood, serum, and plasma specimens segment is expected to dominate the market.
- By site of testing, the laboratory test segment held the largest share of the market in 2025.
- By application, the infectious diseases segment is expected to dominate the market.
- By end user, clinical laboratories segment will grow the fastest during the forecast period.
- F. Hoffmann-La Roche Ltd., Abbott, Danaher Corporation, Sysmex Corporation, Shenzhen Mindray Bio-Medical Electronics Co.,were identified as some of the star players in the Asia Pacific in vitro diagnostics market, supported by their strong market presence and extensive product portfolios.
The fast-growing Asia Pacific in vitro diagnostics market is driven by the expanded improvement in healthcare infrastructure and the rising burden of chronic and lifestyle-related diseases. Many countries are increasingly focusing their efforts on preventive health, which includes utilizing diagnostic services at an earlier stage and promoting the adoption of routine and specialist tests. Additionally, significant diversity in population coupled with incessant improvements in govenmemt supported healthcare initiatives is making diagnosis products readily accessible in care facilities. Collectively, these factors contribute to a steady increase in testing volumes while strengthening the overall market outlook for IVD solutions in Asia Pacific.